Multi Radiance Lasers
The world's most popular therapy laser family. Over 500,000 systems sold worldwide. Patented super-pulsed technology delivers up to 50,000mW peak power with zero thermal risk.




Quick Specs
- Technology
- Super Pulsed
- Wavelengths
- 905nm, 875nm, 660nm
- Peak Power
- 25,000–50,000mW
- Thermal Risk
- None (super pulsed)
- Systems Sold
- 500,000+
- Price Range
- $2,495–$8,995
Trusted super-pulsed systems with up to 50,000mW peak power for deep tissue therapy and pain relief. The MR4 series is one of the most proven therapy laser lines in the world—over 500,000 units sold. FDA cleared for temporary relief of pain, inflammation, and increased circulation.
Choose Your TerraQuant / MR4 System
Two categories: Portable handheld systems for mobility and sports, or Desktop Console systems for clinical volume with upgradeable emitters.
Portable Systems Handheld Super Pulsed
Battery-powered, fully portable super pulsed lasers. Take the world's most effective technology anywhere.
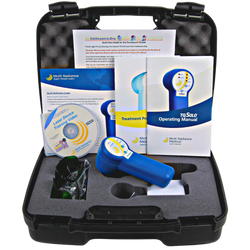
TerraQuant TQ Solo
$2,49525,000mW Peak @905nm • Super Pulsed • Portable
Our most popular system ever. The TQ Solo packs full super-pulsed technology into a compact, battery-powered handheld. Same proven technology as larger systems at an accessible price point.
- Super pulsed 905nm laser
- Infrared SLDs (875nm)
- Red light (660nm)
- Static magnetic field
- Battery powered
- Preset protocols

MR4 Activ
$7,99525,000mW Peak @905nm • Blue Laser • Sports-Focused
Advanced handheld system designed for sports medicine and athletic training. Includes blue laser technology for enhanced surface treatment and antimicrobial effects.
- Super pulsed 905nm laser
- Blue laser included
- Sports-optimized protocols
- Rugged portable design
- Rapid treatment times
- Athletic trainer favorite
MR4 Desktop Consoles Clinical Power Systems
Professional desktop systems with upgradeable emitters, dual-port operation, and maximum treatment flexibility.

MR4 Pro
$6,99525,000mW Peak • Single Emitter
Entry-level desktop console with single 25kW peak power emitter. Upgradeable to 3 emitters as your practice grows.

MR4 LS50
$7,49550,000mW Peak • 4 Laser Diodes
Maximum power desktop with 50kW peak power emitter featuring 4 laser diodes. Larger treatment head covers more area.

MR4 Elite
$8,495Pro + LS50 Emitters • Simultaneous Use
The complete package: includes BOTH the Pro and LS50 emitters. Run both simultaneously for maximum treatment flexibility.

MR4 LaserStim
$8,995Laser + eStim • Skin Conductivity
Combines laser therapy with electrical stimulation. Skin conductivity helps locate problem areas. eStim can be billed separately to insurance.
Compare All TerraQuant / MR4 Systems
| Specification | TQ Solo | MR4 Activ | MR4 Pro | MR4 LS50 | MR4 Elite | MR4 LaserStim |
|---|---|---|---|---|---|---|
| Type | Portable | Portable | Desktop | Desktop | Desktop | Desktop |
| Peak Power | 25,000mW | 25,000mW | 25,000mW | 50,000mW | 25K + 50K | 25,000mW |
| 905nm Laser | ✓ | ✓ | ✓ | ✓ (4 diodes) | ✓ (both) | ✓ |
| 875nm IR SLD | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 660nm Red | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Blue Laser | — | ✓ | — | — | — | — |
| Magnetic Field | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| eStim | — | — | — | — | — | ✓ |
| Skin Conductivity | — | — | — | — | — | ✓ |
| Emitter Ports | — | — | 2 | 2 | 2 | 2 |
| Upgradeable | — | — | ✓ (to 3) | — | — | — |
| Best For | Entry/Home | Sports | Starter Clinic | Power Users | Full Clinic | Insurance Billing |
| Price | $2,495 | $7,995 | $6,995 | $7,495 | $8,495 | $8,995 |
Why TerraQuant / MR4?
Super Pulsed Technology
High power impulses delivered in billionths of a second. A unique approach that minimizes thermal buildup during treatment.
Efficient Delivery
High peak power in nanosecond pulses. Multiradiance's approach to optimizing photon delivery to tissue.
Multiple Radiances
Four technologies working synergistically: super pulsed laser, infrared SLDs, red light, and static magnetic field.
Clinically Proven
Numerous double-blind trials. 88% positive effect demonstrated for arthritis pain. Over 4,000 studies validate laser therapy.
Turnkey Systems
Easy to set up and use. Preset protocols for common conditions. No complicated programming required.
500,000+ Systems Sold
The world's largest therapy laser company. Proven in 30+ countries. The industry standard worldwide.
Questions About TerraQuant / MR4?
Free consultation available. We'll help you choose the right system for your practice or home use.
The World's Most Popular Therapy Laser Technology
With over 500,000 systems sold worldwide, Multiradiance has quietly become the largest therapy laser company in the world. Their TerraQuant and MR4 systems are used in clinics across 30+ countries, establishing super-pulsed technology as a widely-adopted approach to photobiomodulation therapy.
What makes them different? While most therapy lasers use continuous wave output, TerraQuant/MR4 systems use super-pulsing—delivering high peak power (up to 50,000mW) in ultra-short bursts measured in billionths of a second. According to Multiradiance, this approach optimizes photon delivery while minimizing thermal buildup in tissue.
This unique pulsing methodology has made TerraQuant/MR4 a popular choice for practitioners who prefer the super-pulsed approach to laser therapy.
Understanding Super Pulsed Technology
Up to 50,000mW in each pulse, delivered in billionths of a second rather than continuous output.
Multiradiance claims high peak power creates concentrated photon density for effective treatment.
Short pulse duration allows tissue to dissipate energy between pulses, reducing heat accumulation.
Four Radiances Working Together
TerraQuant and MR4 systems don't rely on laser alone. They combine four therapeutic technologies that work synergistically to create the optimal biological environment for healing.
The core technology. High peak power (25,000–50,000mW) delivered in nanosecond pulses. According to Multiradiance, this approach optimizes photon delivery while the pulsing minimizes thermal effects. This is where the "heavy lifting" happens.
Superluminous diodes provide broader spectrum coverage than laser alone. Gently heats the surface tissue layer, improving circulation and preparing tissue for laser treatment.
Red light targets surface tissue with proven anti-inflammatory effects. Accelerates wound healing, reduces swelling, and complements the infrared wavelengths.
The magnetic field helps maintain the ionic channel environment within cells, potentially enhancing the cellular response to light therapy and supporting the overall healing process.
Together, these four radiances are designed to create comprehensive treatment coverage through complementary wavelengths and technologies.
Understanding Peak Power vs. Average Power
It's important to understand that peak power (shown below) is different from average power. Super pulsed lasers deliver high power in very short bursts, but the average power over time is much lower than continuous wave Class IV systems. Both approaches have their advocates.
Note: Super pulsed systems measure peak power during each nanosecond pulse. Class IV and Class 3B systems measure continuous or average power. These are different metrics—higher peak power does not necessarily mean more total energy delivered. Each technology has distinct advantages depending on your treatment philosophy and clinical goals.
Clinical Evidence
Demonstrated efficacy in double-blind studies for arthritis pain in small and medium joints.
Laser therapy is one of the most researched modalities in physical medicine.
TerraQuant/MR4 systems are the standard of care across the globe.
Numerous randomized double-blind clinical trials have validated the efficacy of TerraQuant and MR4 systems for various health conditions. Unlike many therapy devices, these results come from rigorous scientific methodology—not just anecdotal reports.
Clinical Applications
Pain Conditions
- Acute and chronic pain
- Back pain / Neck pain
- Arthritis (all types)
- Fibromyalgia
- Neuropathy
- Carpal tunnel syndrome
- Post-surgical pain
Musculoskeletal
- Sports injuries
- Muscle strain
- Tendinitis
- Bursitis
- Tennis elbow
- Plantar fasciitis
- Joint injuries
Healing & Recovery
- Wound healing
- Post-operative recovery
- Soft tissue repair
- Edema reduction
- Inflammation
- Circulation improvement
- Athletic recovery
How Laser Therapy Works at the Cellular Level
Photons from the laser are absorbed by chromophores in the mitochondria (the cell's "power plants").
Absorbed light energy stimulates cytochrome c oxidase, accelerating ATP (cellular energy) production.
Increased ATP transforms damaged cells from a state of injury to stable, healthy function.
The biochemical increase in cellular energy is what enables accelerated healing. Cells with more energy can repair faster, reduce inflammation more efficiently, and restore normal function more quickly.
Which System Is Right For You?
For Home Users / Entry Level
Full super-pulsed technology in a portable package. Same proven results as larger systems. Our most popular system ever for good reason.
For Sports Medicine / Athletic Trainers
Handheld portability with blue laser for antimicrobial applications. Sports-optimized protocols. Take it anywhere athletes need treatment.
For New Practices / Budget-Conscious
Desktop console with upgrade path. Start with one emitter, add up to two more as your practice grows. Lowest desktop entry point.
For Maximum Power
50,000mW peak power with 4 laser diodes. Larger treatment head. When you need the most power and fastest treatments.
For Complete Clinics
Both Pro and LS50 emitters included. Run simultaneously. Maximum flexibility for any condition. Best overall value for serious practices.
For Insurance Billing / Diagnostics
Laser + eStim in one device. Skin conductivity helps locate problem areas. Bill eStim as separate line item to insurance companies.
Comparing Laser Therapy Approaches
Different laser technologies take different approaches to photobiomodulation. Here's how they compare:
| Feature | Super Pulsed (TQ/MR4) | Class IV (Continuous) | Class 3B (CW) |
|---|---|---|---|
| Peak Power | 25,000–50,000mW | 10,000–60,000mW | 500–2,700mW |
| Average Power | Lower (pulsed) | High (continuous) | Low–Medium |
| Delivery Method | Nanosecond pulses | Continuous wave | Continuous wave |
| Thermal Considerations | Minimal buildup | Requires monitoring | Minimal |
| Multiple Wavelengths | Yes (4 radiances) | Usually 1–2 | 1–2 |
| Portability | Available | Limited | Excellent |
| Treatment Speed | Moderate | Fast | Slower |
| Price Range | $2,495–$8,995 | $8,000–$55,000+ | $2,000–$9,000 |
Each technology has its proponents. Super pulsed advocates emphasize the high peak power and pulsing methodology. Class IV advocates emphasize total energy delivery and treatment speed. Many practitioners use both depending on the application.
FDA-Cleared Indications
TerraQuant and MR4 systems are FDA-cleared for pain relief and healing through patented multiple therapeutic radiances:
Frequently Asked Questions
Class IV continuous lasers deliver constant output (typically 10,000–60,000mW average power). This provides high total energy delivery for fast treatments, though thermal management is important.
Super pulsed lasers (TerraQuant/MR4) deliver high peak power (up to 50,000mW) but in ultra-short bursts—billionths of a second. The average power over time is lower, but Multiradiance claims the pulsing methodology offers therapeutic advantages.
Both technologies have strong clinical followings. Many practitioners choose based on their treatment philosophy, patient volume, and specific applications. Some practices use both types for different situations.
TQ Solo ($2,495) – Best for home users, entry-level practitioners, or anyone who values portability. Full technology at the lowest price.
MR4 Activ ($7,995) – Best for sports medicine, athletic trainers, or practitioners who need portability plus blue laser technology.
MR4 Pro ($6,995) – Best for new practices with growth plans. Desktop with upgrade path to 3 emitters.
MR4 LS50 ($7,495) – Best for practices that want maximum power. 50kW peak with larger treatment head.
MR4 Elite ($8,495) – Best overall value for established practices. Includes both emitters for any application.
MR4 LaserStim ($8,995) – Best for practices that bill insurance for eStim or want diagnostic point-finding capability.
Yes. The TQ Solo uses the same super-pulsed technology at the same 25,000mW peak power as the MR4 Pro desktop. It includes all four radiances: super pulsed laser, infrared SLDs, red light, and magnetic field.
The main differences with desktop systems are: (1) the LS50 has 50,000mW peak power versus 25,000mW; (2) desktop consoles have dual ports for running two emitters simultaneously; (3) desktop systems are better suited for high-volume clinical use.
For home users and many practitioners, the TQ Solo delivers identical therapeutic benefit at a fraction of the cost.
Super pulsed means the laser fires in extremely short bursts—typically 100–200 nanoseconds (billionths of a second). During each pulse, the peak power is high (25,000–50,000mW), but the pulse is very brief.
Multiradiance claims this creates high "photon density"—a concentrated burst of light energy during each pulse. They state this pulsing methodology optimizes energy delivery while the brief pulse duration minimizes thermal accumulation.
Between pulses, tissue has time to dissipate energy before the next pulse arrives. This "duty cycle" is central to the super pulsed approach to laser therapy.
Typical treatments range from 3–10 minutes depending on the condition and treatment area. The preset protocols on the device calculate optimal treatment times automatically based on the selected condition.
Because of the high peak power and efficient delivery, TerraQuant/MR4 treatments are often completed quickly—typically achieving results in minutes.
Different wavelengths and technologies have different characteristics and biological effects:
905nm super pulsed laser – The primary therapeutic wavelength. High peak power for ATP production and pain reduction.
875nm infrared SLDs – Broader spectrum coverage. Improves circulation and prepares tissue.
660nm red light – Surface-level anti-inflammatory effects. Accelerates wound healing.
Static magnetic field – Maintains cellular ionic environment. Supports overall healing response.
Working together, these four technologies create a more complete therapeutic effect than any single modality alone—treating from skin surface to deep tissue simultaneously.
The LaserStim adds two features on top of standard laser therapy:
Skin conductivity detection – The device can measure changes in skin conductivity to help locate problem areas, trigger points, or areas of inflammation. This takes guesswork out of treatment positioning.
Electrical stimulation (eStim) – Delivers eStim simultaneously with laser therapy. This is significant for insurance billing because many insurers will reimburse eStim as a separate line item from laser therapy. One treatment session, two billable modalities.
For practices focused on maximizing insurance reimbursement, the LaserStim can pay for itself through the additional billing codes.
Yes. TerraQuant/MR4 accessories expand your treatment capabilities:
Available accessories include pinpoint probes for precise trigger point work, large treatment emitters for covering bigger areas, and various specialized attachments. The MR4 Pro can be upgraded from one emitter to three emitters over time.
The desktop consoles have two ports, allowing simultaneous use of two emitters—treat multiple sites at once or use different emitter types on the same patient.
Ready to Experience TerraQuant / MR4?
Join the 500,000+ practitioners and patients worldwide who trust the world's most proven therapy laser technology.